 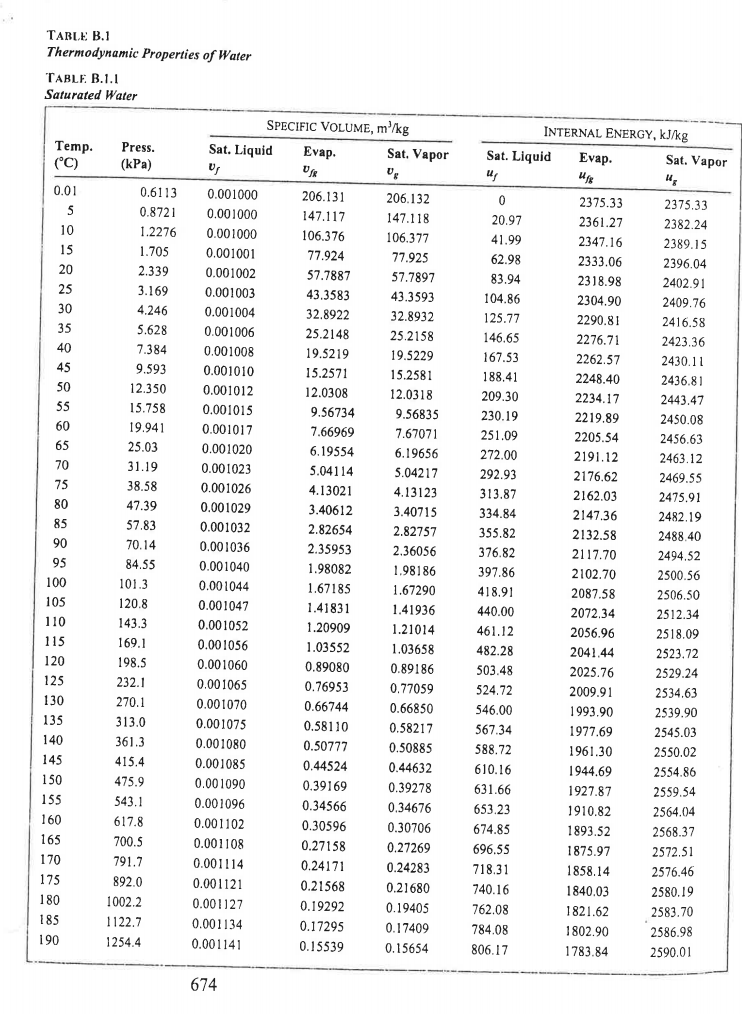
This is why seawater at Antarctica is very salty. The salt that is rejected forms brine beneath the ice and becomes more and more salty until it becomes so dense that it sinks, displaces less dense seawater that moves to the surface. In everyday life, it is often assumed to approximate the water density to 1000 k g. in the so-called normal conditions), the water density is equal to 1000 k g / m 3 kg/m3 k g / m 3. Both below and above this temperature, the density of water is lower.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
